Bullous pemphigoid (BP) is considered the most frequent autoimmune blistering disease with an incidence of about 1.5 to 2 per 100,000 inhabitants and year in the general population. The disease becomes even more frequent in the elderly. The clinical picture of BP includes tense blisters, formation of erythema, erosions, crusts and pruritus. The presence of autoantibodies against two hemidesmosomal proteins called BP180 and BP230, which are responsible for the anchorage of the epidermal cells to the basal lamina, is an immunological characteristic of the autoimmune disease.
About 80% to 90% of BP patients form antibodies against the immunodominant non-collagenous domain 16 (NC16A) of BP180, often along with antibodies against other BP epitopes. A pathogenic role of the anti-BP180NC16A antibodies has been demonstrated by several experiments – to remove them by immunoadsorption therefore appears to be a reasonable therapeutic option. Applying this technique, immunoglobulins and immune complexes are bound by a matrix and are thus depleted from the patient’s blood circulation.
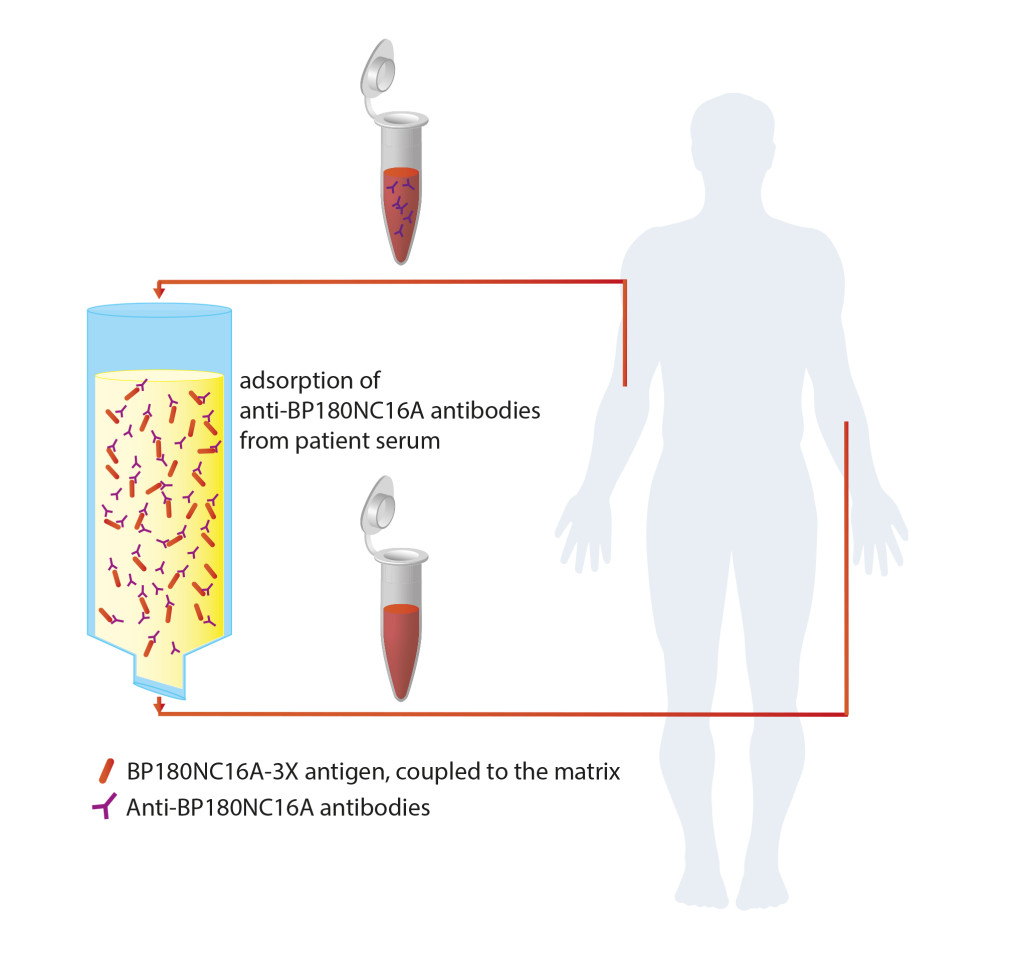
A new immune adsorber, consisting of a matrix and a coupled BP180NC16A-3X antigen, was now presented in a novel publication by Mersmann et al. It specifically binds antibodies targeting BP180NC16A and has been developed for the treatment of patients with severe and/or refractory BP. The measurement of the antibody concentration concentration in sera of 9 BP patients prior to and after the adsorption using the Anti-BP180NC16A-4X ELISA revealed an average reduction of the antibody level by 98%. To assess possible unspecific adsorption, levels of antibodies against measles and BP230 were determined. Their quantity was reduced by only 5% and 9%, respectively, which points to the high specificity of the adsorber for anti-BP180 antibodies.
Twelve consecutive cycles of adsorption and subsequent regeneration (antibody elution) of the BP180NC16A-3X matrix led to a just minimal decline of the antibody binding capacity of 5%. Thus, the antigen stays intact during regeneration of the matrix and the adsorber can be applied for several adsorption cycles. Furthermore the BP180NC16A-3X matrix is autoclavable without impairment of its binding efficiency. Sterility is a prerequisite for its clinical application.
Thus, immunoadsorption using the BP180NC16A-3X adsorber may provide a valuable alternative therapeutic option for BP patients. Up to now, they are usually treated with strong immunosuppressive medication which is often associated with severe adverse effects, in particular in older patients. Disadvantages of the currently established generalised immunoadsorption such as the increased risk of secondary infections due to the unspecific depletion of protective antibodies, is countervailed by the specificity of the adsorber for anti-BP180NC16A antibodies. Especially during the initial treatment phase of BP, the novel adsorber may be of great importance to rapidly decrease the autoantibody concentration.