Press release
Luebeck, 29th January 2016
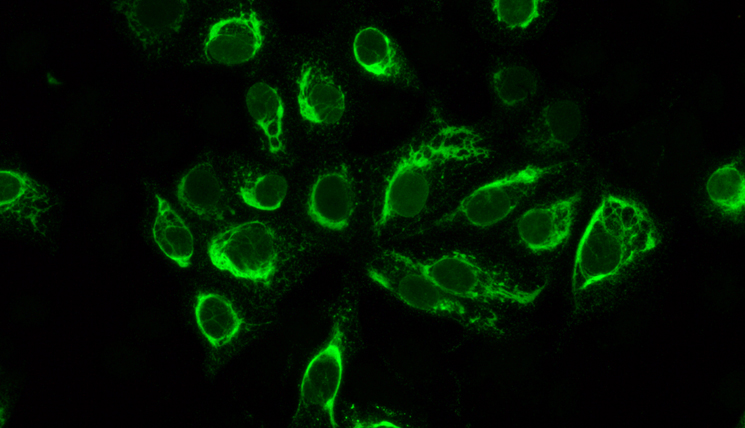
EUROIMMUN AG, a manufacturer of test systems and instruments for medical diagnostics with headquarters in Luebeck, Germany, has developed the first complete test package worldwide for the serological detection of Zika virus infections. The ELISA and indirect immunofluorescence tests allow the determination of specific antibodies (IgM, IgG) against the virus in the blood of infected patients.
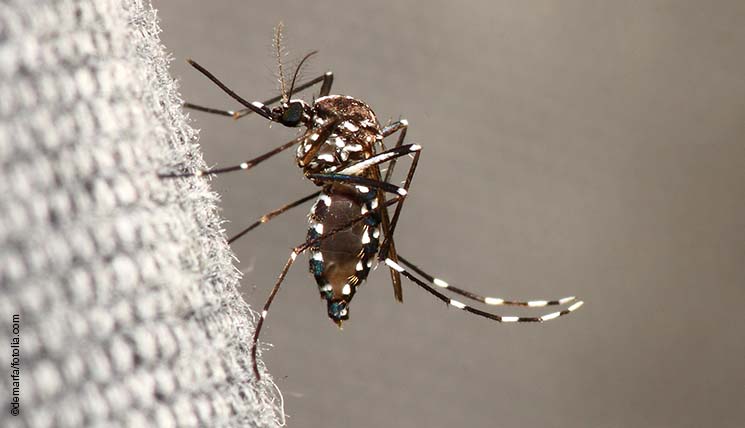
Zika virus was first identified in Uganda in 1947. It is transmitted to humans by mosquitoes of the Aedes species. Infections with the virus manifest with mild fever, headaches, rash and conjunctivitis. Around 80% of infections remain unnoticed by the patient.
The virus has been spreading over the American continent since the beginning of 2015, with Brazil being most severely affected. Since October, health authorities in the affected Brazilian regions have been reporting a more than twentyfold increase in the incidence of microcephaly cases. Microcephaly refers to a developmental disorder of the head and brain in newborns. Moreover, an increased number of patients with a rare neurological disease, the Guillain-Barré syndrome (GBS), was reported during a large Zika virus outbreak in French Polynesia in 2013/2014. Brazil and El Salvador have similarly reported conspicuous numbers of GBS cases during the current Zika epidemic in Latin America. The question whether there is a causal link between the Zika virus infections and the foetal malformations and cases of GBS in addition to the temporal and spatial association is now subject of intense research.
The EUROIMMUN tests are now available for the diagnosis of Zika virus infections and will shortly be used in studies on the subject at the Fiocruz (Fundação Oswaldo Cruz), an institute linked to the Brazilian health ministry. “We have reacted immediately to the acute demand and developed ELISA and indirect immunofluorescence tests which we have evaluated in cooperation with the Bernhard-Nocht Institute in Hamburg. The tests are suitable for the fast screening of large sample numbers and therefore enable monitoring of the virus spread,” says Katja Steinhagen, head of the ELISA Infectious Serology department at EUROIMMUN AG.
The antibody tests from EUROIMMUN can detect both acute infections (in most cases from the fifth day after onset of symptoms) and past infections with Zika virus. Test systems for the direct detection of the virus (e.g. PCR) are only able to detect infections up to 5 days after onset of symptoms – after this, the virus itself is no longer detectable.
“Our ELISAs are based on very specific, recombinant antigens which are produced in our own research laboratories. Positive results are a clear indicator of a Zika virus infection” says Katja Steinhagen. “And our special immunofluorescence mosaics help laboratory physicians to investigate patient samples for other viral fever diseases in parallel. This is important for differential diagnosis, since in Latin America there are also dengue and chikungunya viruses, which cause similar symptoms and are transmitted by the same species of mosquitoes as the Zika virus. We hope our test systems can help to clarify as soon as possible whether there is a link between the cases of microcephaly and Zika infections”.
Visit www.zika-diagnostics.com for more information.




Dear Sir or Madame,
We, at the center for Autoimmune Diseases Research (CREA) at the Universidad del Rosario in Bogota, Colombia, South America, would like to urgently order some Kits Anti-Zika Virus ELISA (IgG/IgM) # EI 2668-9601 G/M and IIFT Arboviral Fever Mosaic 2 (IgG/IgM).
Thank you for your prompt attention.
Dear Mr Anaya,
thank you for your message. I forwarded your request to the person in charge and he will contact you immediately.
Best regards,
Johanna