There is a huge variety of neurological diseases and it often takes specialists a long time to figure out the cause of a patient’s symptoms. Doctors regularly encounter patients with unspecific neurological complaints. In these cases, differential diagnostics is crucial to finding the right therapy. Even if the doctor has a suspicion and there are signs pointing in a certain direction, result constellations are not always clear and specific enough to allow for a definite diagnosis. This means that all the diagnostic results need to be considered together to allow for a plausible conclusion. This was the case for Elisabeth:
The 73-year-old from Frankfurt* consulted her general practitioner for back pain and headache at night with mild facial paralysis, general fatigue and an episode of mild fever. In the consultation Elisabeth told her doctor that she normally felt healthy and was therefore still very active in her everyday life, often going for walks in the woods and tending to her large garden. Elisabeth’s frequent outdoor activities and her symptoms raised the initial suspicion that she might have been infected with Borrelia via an unnoticed tick bite and might have developed neuroborreliosis. Her blood (serum) and cerebrospinal fluid (CSF) were collected for laboratory diagnostic investigation.
In the visual assessment, the CSF sample looked clear and normal. However, the cell count was found to be increased. This so-called pleocytosis is a first indication of an acute inflammatory process of the central nervous system (CNS). In addition to the cell count, the protein levels in CSF and serum were measured. Based on the determination of the albumin quotient, the Reiber diagramme revealed a blood-CSF barrier dysfunction, which indicates an illness-related protein accumulation in the CSF. It is a typical finding in neuroborreliosis but is non-specific as it can also occur in other diseases with neurological symptoms. Furthermore, an intrathecal synthesis of total IgM but not of total IgG could be observed. This IgM dominance in the Reiber diagramme is yet another indication of neuroborreliosis. In the determination of oligoclonal bands, more antibody bands were detected in the CSF than in the serum sample. Oligoclonal bands in the CSF typically occur in neuroborreliosis, but may also be found in other inflammatory diseases of the CNS (e.g. multiple sclerosis).
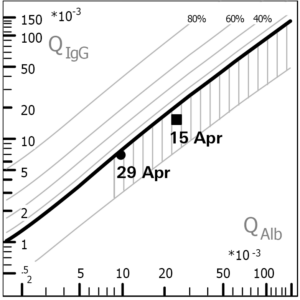
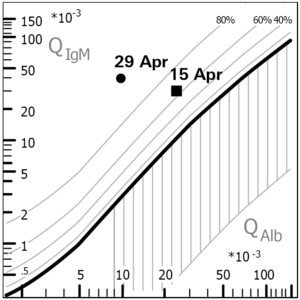
To further confirm the suspected diagnosis, a value for the pathogen-specific antibody production in the CSF was determined: the antibody index (AI). It was calculated using the results of the Anti-Borrelia ELISAs (IgM and IgG) for the serum/CSF pair. Unexpectedly, a normal AI was calculated for IgM. However, the IgM antibodies in the serum sample indicated an acute Borrelia infection. For IgG, no AI could be calculated, because no anti-Borrelia IgG antibodies could be detected in the serum sample taken at the same time as the CSF sample, so there was no reference value for calculating the AI. As IgG antibodies are usually not produced until several weeks after infection, it is possible that no AI can be calculated in the early phases of an infection.
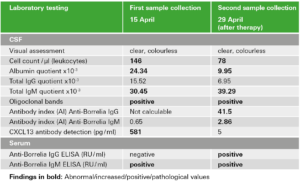
Due to the unusual and inconclusive result constellation, the determination of chemokine CXCL13 in the CSF was advisable. The concentration of this signalling protein was significantly increased in Elisabeth’s sample. This confirmed the suspicion of acute neuroborreliosis, as CXCL13 levels are especially high in acute, untreated cases. Furthermore, CXCL13 is a relevant marker for the evaluation of therapy success as can be seen by the decreased concentration in the follow-up sample of the patient.
After two weeks of antibiotic treatment, the cell count and the CXCL13 concentration in the CSF had reduced and the blood-CSF barrier dysfunction had receded, both of which indicate successful therapy. In retrospect, the diagnosis of neuroborreliosis could also be clearly confirmed by laboratory diagnostic findings in a follow-up sample (IgG seroconversion, pathological AI). Elisabeth’s case represents an individual result constellation.
There are a variety of possible combinations of indications that must always be considered together to allow for diagnosis of a neurological disease. For example, when no infectious cause is found for acute neurological symptoms, further differential diagnostics may focus on the analysis of autoantibodies against neuronal target antigens. As a concrete example, this may allow autoimmune encephalitis to be distinguished from CNS infections, which is crucial due to the very different treatment of autoimmune- and infection-related neurological diseases.
EUROIMMUN offers a unique portfolio of test systems for neurological disease diagnostics as well as flexible automation solutions and a comprehensive service for laboratories. Discover our spectrum of analysis parameters for the areas of CNS infections, neuronal autoimmunity and neurodegeneration: www.neuro-company.com
*The name and personal details were changed by the editors.
Intrathecal antibodies: Antibodies produced in the CNS.
Serum/CSF pair: CSF and serum must be collected and tested simultaneously as isolated antibody results for the CSF without reference to the individual levels in the blood are not conclusive due to the fact that all proteins in the blood may enter the CSF by diffusion.
Albumin quotient: Albumin is a protein that–in contrast to antibodies–is never produced in the CNS; it can only enter the CNS by diffusion from the blood. For this reason, the calculated quotient of the albumin concentration in the CSF and in the serum is a suitable means to determine the diffusion-related distribution of proteins (e.g. antibodies) and detect changes caused by diseases such as a blood-CSF barrier dysfunction.
Blood-CSF barrier dysfunction: A blood-CSF barrier dysfunction, which is characterised by an increased albumin quotient (QAlb), is caused by a disease-related reduction in the CSF flow rate, which may lead to an accumulation of molecules/proteins in the CSF.
Reiber diagramme: In the CSF/serum quotient diagramme developed by Professor Hansotto Reiber the immunoglobulin quotient (QIg for IgG, IgM or IgA) is plotted against the albumin quotient (QAlb). Based on certain reference values, the graphical representation allows to identify a barrier dysfunction or intrathecal antibody production at a glance. Their isolated or combined presence may be an indication of an underlying disease.
Antibody index (AI): The antibody index is a value for the identification of an additional production of specific antibodies against an infectious agent in the CSF (intrathecal antibody synthesis). To be able to distinguish between diffused and intrathecally produced antibodies, the pathogen-specific CSF/serum antibody quotient is brought into relation with the CSF/serum quotient of the respective total immunoglobulin (IgG, IgM or IgA). If the pathogen-specific quotient is greater than the total immunoglobulin quotient (AI > 1.5), a pathogen-specific antibody synthesis in the CNS is presumed.
Oligoclonal bands: In isoelectric focusing, antibodies are separated in an electric field along a pH gradient. They migrate to different lengths (up to their isoelectric point) depending on their net charge and can be seen as bands after staining. If antibody bands are seen in the CSF fraction that do not appear in the serum, an intrathecal synthesis and thus an inflammatory reaction in the CNS is likely.
Chemokines: Chemokines are chemotactic cytokines, i. e. signalling proteins, that direct different immune cells to a site of inflammation.

